- What is Ozone?
- What is a Dobson Unit?
- What is the Ozone Hole?
- History of the Ozone Hole
- What is EESC?
Topics
What is Ozone?
Ozone is a gas made up of three oxygen atoms (O3). It occurs naturally in small (trace) amounts in the upper atmosphere (the stratosphere). Ozone protects life on Earth from the Sun’s ultraviolet (UV) radiation. In the lower atmosphere (the troposphere) near the Earth’s surface, ozone is created by chemical reactions between air pollutants from vehicle exhaust, gasoline vapors, and other emissions. At ground level, high concentrations of ozone are toxic to people and plants.
Stratospheric “good” ozone
Ninety percent of the ozone in the atmosphere sits in the stratosphere, the layer of atmosphere between about 10 and 50 kilometers altitude. The natural level of ozone in the stratosphere is a result of a balance between sunlight that creates ozone and chemical reactions that destroy it. Ozone is created when the kind of oxygen we breathe—O2—is split apart by sunlight into single oxygen atoms. Single oxygen atoms can re-join to make O2, or they can join with O2 molecules to make ozone (O3). Ozone is destroyed when it reacts with molecules containing nitrogen, hydrogen, chlorine, or bromine. Some of the molecules that destroy ozone occur naturally, but people have created others.
The total mass of ozone in the atmosphere is about 3 billion metric tons. That may seem like a lot, but it is only 0.00006 percent of the atmosphere. The peak concentration of ozone occurs at an altitude of roughly 32 kilometers (20 miles) above the surface of the Earth. At that altitude, ozone concentration can be as high as 15 parts per million (0.0015 percent).
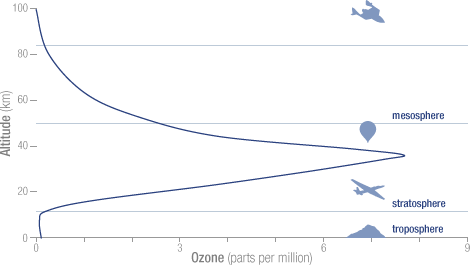
The concentration of ozone varies with altitude. Peak concentrations, an average of 8 molecules of ozone per million molecules in the atmosphere, occur between an altitude of 30 and 35 kilometers.
Ozone in the stratosphere absorbs most of the ultraviolet radiation from the Sun. Without ozone, the Sun’s intense UV radiation would sterilize the Earth’s surface. Ozone screens all of the most energetic, UV-c, radiation, and most of the UV-b radiation. Ozone only screens about half of the UV-a radiation. Excessive UV-b and UV-a radiation can cause sunburn and can lead to skin cancer and eye damage.
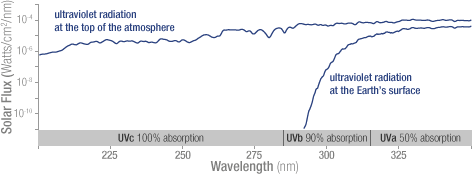
Solar ultraviolet radiation is largely absorbed by the ozone in the atmosphere—especially the harmful, high-energy UV-a and UV-b. The graph shows the flux (amount of energy flowing through an area) of solar ultraviolet radiation at the top of the atmosphere (top line) and at the Earth’s surface (lower line). The flux is shown on a logarithmic scale, so each tick mark on the y-axis indicates 10 times more energy.
Increased levels of human-produced gases such as CFCs (chlorofluorocarbons) have led to increased rates of ozone destruction, upsetting the natural balance of ozone and leading to reduced stratospheric ozone levels. These reduced ozone levels have increased the amount of harmful ultraviolet radiation reaching the Earth’s surface. When scientists talk about the ozone hole, they are talking about the destruction of stratospheric, “good,” ozone.
Tropospheric “bad” ozone
Although ozone high up in the stratosphere provides a shield to protect life on Earth, direct contact with ozone is harmful to both plants and animals (including humans). Ground-level, “bad,” ozone forms when nitrogen oxide gases from vehicle and industrial emissions react with volatile organic compounds (carbon-containing chemicals that evaporate easily into the air, such as paint thinners). In the troposphere near the Earth’s surface, the natural concentration of ozone is about 10 parts per billion (0.000001 percent). According to the Environmental Protection Agency, exposure to ozone levels of greater than 70 parts per billion for 8 hours or longer is unhealthy[1]. Such concentrations occur in or near cities during periods where the atmosphere is warm and stable. The harmful effects can include throat and lung irritation or aggravation of asthma or emphysema.
The following link will take you to a non-NASA site.
[1] 2015 National Ambient Air Quality Standards (NAAQS) for Ozone.

